Introduction: Classic Hodgkin lymphoma (CHL) features a unique crosstalk between malignant cells and different types of normal immune cells in the tumor-microenvironment (TME). On the basis of histomorphologic and immunophenotypic features of the malignant Hodgkin and Reed-Sternberg (HRS) cells and infiltrating immune cells, four histological subtypes of CHL are recognized: Nodular sclerosing (NS), Mixed cellularity, Lymphocyte-rich (LR) and Lymphocyte-depleted CHL. Recently, our group described the high abundance of various types of immunosuppressive CD4+ T cells including LAG3+ and/or CTLA4+ cells in the TME of CHL using single cell RNA sequencing (scRNAseq). However, the TME of LR-CHL has not been well characterized due to the rarity of the disease. In this study, we aimed at characterizing the immune cell profile of LR-CHL at single cell resolution.
METHODS: We performed scRNAseq on cell suspensions collected from lymph nodes of 28 primary CHL patients, including 11 NS, 9 MC and 8 LR samples, with 5 reactive lymph nodes (RLN) serving as normal controls. We merged the expression data from all cells (CHL and RLN) and performed batch correction and normalization. We also performed single- and multi-color immunohistochemistry (IHC) on tissue microarray (TMA) slides from the same patients. In addition, an independent validation cohort of 31 pre-treatment LR-CHL samples assembled on a TMA, were also evaluated by IHC.
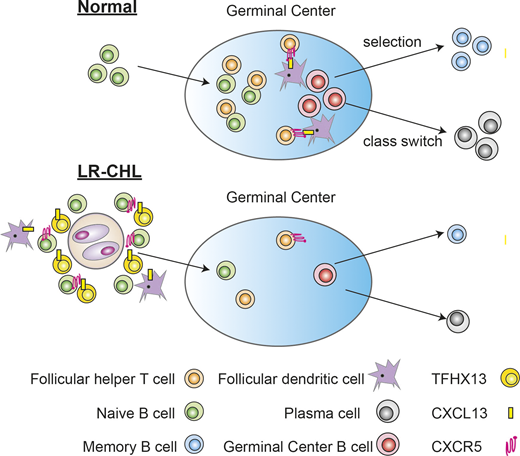
Results: A total of 23 phenotypic cell clusters were identified using unsupervised clustering (PhenoGraph). We assigned each cluster to a cell type based on the expression of genes described in published transcriptome data of sorted immune cells and known canonical markers. While most immune cell phenotypes were present in all pathological subtypes, we observed a lower abundance of regulatory T cells (Tregs) in LR-CHL in comparison to the other CHL subtypes. Conversely, we found that B cells were enriched in LR-CHL when compared to the other subtypes and specifically, all four naïve B-cell clusters were quantitatively dominated by cells derived from the LR-CHL samples.
T follicular helper (TFH) cells support antibody response and differentiation of B cells. Our data show the preferential enrichment of TFH in LR-CHL as compared to other CHL subtypes, but TFH cells were still less frequent compared to RLN. Of note, Chemokine C-X-C motif ligand 13 (CXCL13) was identified as the most up-regulated gene in LR compared to RLN. CXCL13, which is a ligand of C-X-C motif receptor 5 (CXCR5) is well known as a B-cell attractant via the CXCR5-CXCL13 axis. Analyzing co-expression patterns on the single cell level revealed that the majority of CXCL13+ T cells co-expressed PD-1 and ICOS, which is known as a universal TFH marker, but co-expression of CXCR5, another common TFH marker, was variable. Notably, classical TFH cells co-expressing CXCR5 and PD-1 were significantly enriched in RLN, whereas PD-1+ CXCL13+ CXCR5- CD4+ T cells were significantly enriched in LR-CHL. These co-expression patterns were validated using flow cytometry. Moreover, the expression of CXCR5 on naïve B cells in the TME was increased in LR-CHL compared to the other CHL subtypes
We next sought to understand the spatial relationship between CXCL13+ T cells and malignant HRS cells. IHC of all cases revealed that CXCL13+ T cells were significantly enriched in the LR-CHL TME compared to other subtypes of CHL, and 46% of the LR-CHL cases showed CXCL13+ T cell rosettes closely surrounding HRS cells. Since PD-1+ T cell rosettes are known as a specific feature of LR-CHL, we confirmed co-expression of PD-1 in the rosetting cells by IHC in these cases.
Conclusions: Our results reveal a unique TME composition in LR-CHL. LR-CHL seems to be distinctly characterized among the CHL subtypes by enrichment of CXCR5+ naïve B cells and CD4+ CXCL13+ PD-1+ T cells, indicating the importance of the CXCR5-CXCL13 axis in the pathogenesis of LR-CHL.
Savage:BeiGene: Other: Steering Committee; Merck, BMS, Seattle Genetics, Gilead, AstraZeneca, AbbVie: Honoraria; Roche (institutional): Research Funding; Merck, BMS, Seattle Genetics, Gilead, AstraZeneca, AbbVie, Servier: Consultancy. Scott:Janssen: Consultancy, Research Funding; Celgene: Consultancy; NanoString: Patents & Royalties: Named inventor on a patent licensed to NanoString, Research Funding; NIH: Consultancy, Other: Co-inventor on a patent related to the MCL35 assay filed at the National Institutes of Health, United States of America.; Roche/Genentech: Research Funding; Abbvie: Consultancy; AstraZeneca: Consultancy. Steidl:AbbVie: Consultancy; Roche: Consultancy; Curis Inc: Consultancy; Juno Therapeutics: Consultancy; Bayer: Consultancy; Seattle Genetics: Consultancy; Bristol-Myers Squibb: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.